Customer Focus
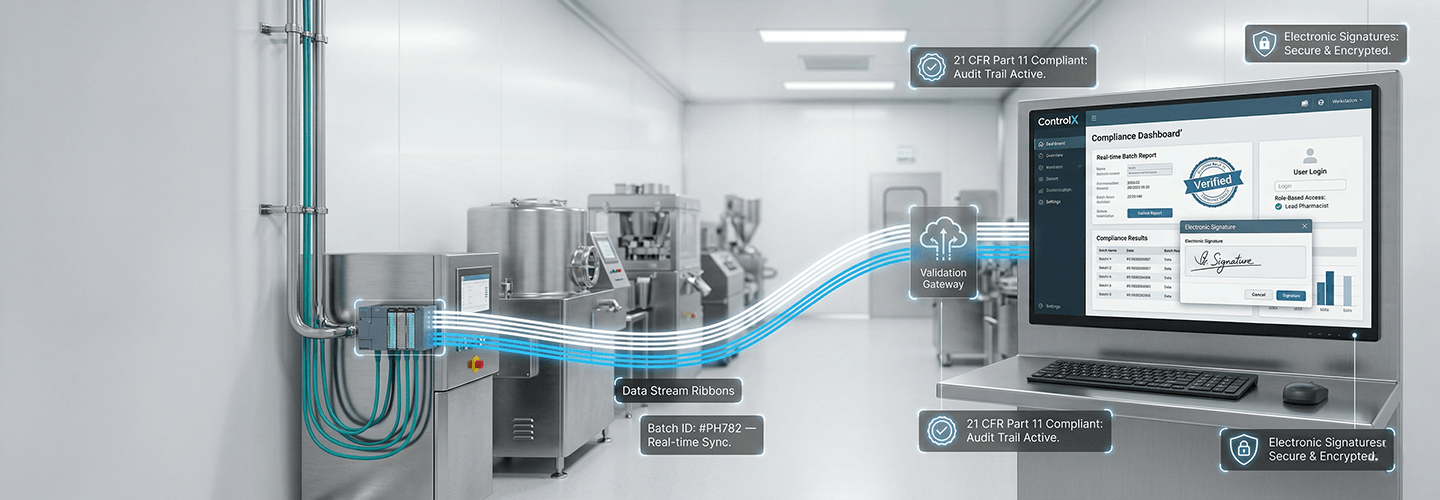
A leading pharmaceutical manufacturer operating granulation, coating, and compression lines required higher data integrity and audit readiness. With increasing regulatory pressure and complex batch processes, the focus shifted toward digital traceability, centralized visibility, and secure data management. The objective was to strengthen compliance while improving process transparency and operational control across the plant.
Requirement
The customer needed a centralized SCADA platform to connect existing PLC based machines and enable plant wide monitoring. The system had to support secure data logging, batch wise recording, audit trails, and automated reporting. User authentication, role based access, and electronic signatures were essential to meet regulatory compliance and ensure reliable process documentation.